ETHICS SELF-CHECK APPLICATION FOR IRB APPROVAL
ETHICS SELF-CHECK APPLICATION FOR IRB APPROVAL.
ETHICS SELF-CHECK APPLICATION FOR IRB APPROVAL
Save your time - order a paper!
Get your paper written from scratch within the tight deadline. Our service is a reliable solution to all your troubles. Place an order on any task and we will take care of it. You won’t have to worry about the quality and deadlines
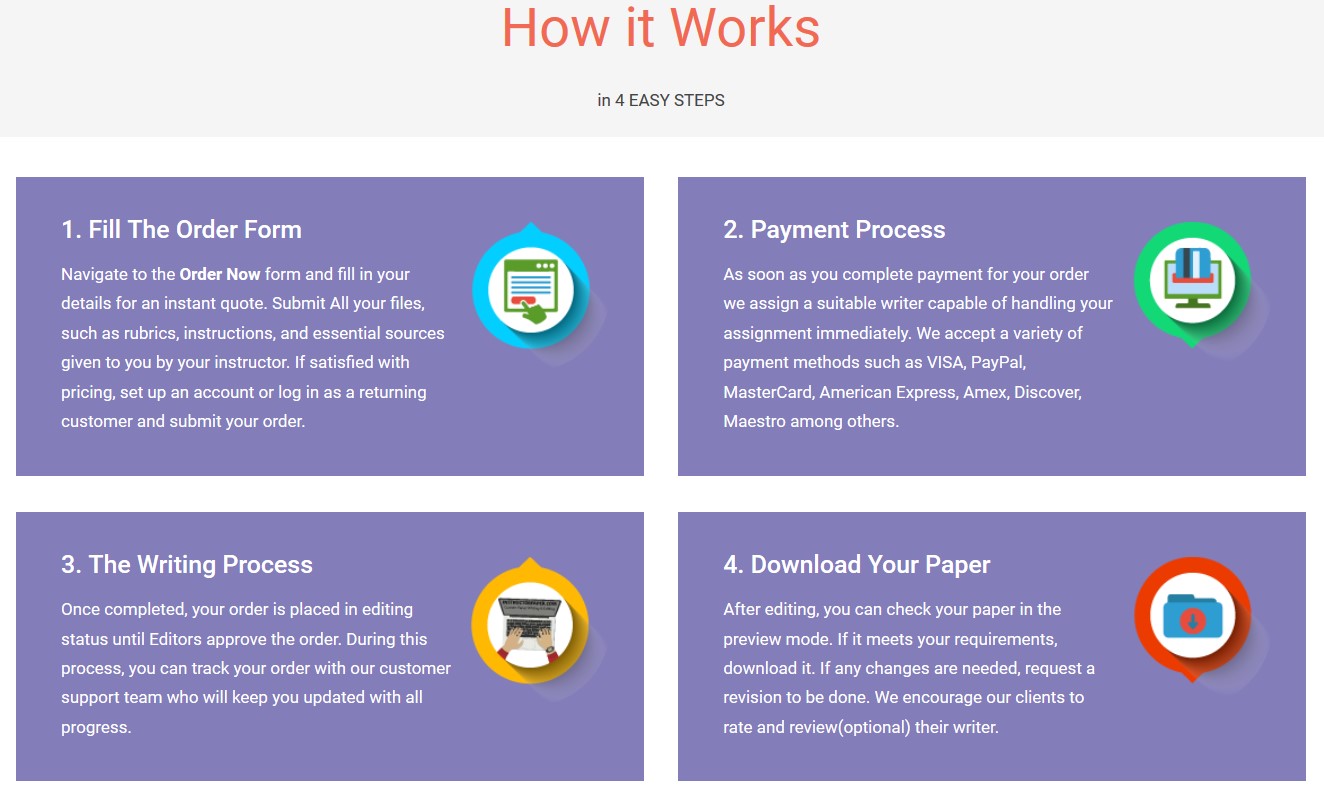
Order Paper NowINSTRUCTIONS:
Section 1: The researcher must complete the brown column A of the table below to document how the research procedures comply with the university’s 40 ethical standards. Mark “Not Applicable” only when there is no possible way to address that ethical issue.
Section 2: Attach enclosures as indicated in section II (yellow section).
Section 3: Provide electronic signature.
Section 4 (students only): Have your faculty supervisor review the entire form and then provide an electronic signature.
IRB approval will be issued when the IRB confirms that there is adequate evidence that the university’s ethical standards have been met, based on this form and the attachments listed in Section II of this form. Within 10 business days of receiving a researcher’s submission, the IRB will notify the researcher of one of the following outcomes:
(a) that the IRB has provided ethics approval based on the submitted documents; or
(b) that the IRB requires revisions and/or additional documentation (will be specified in Column B).
| SECTION I: RESEARCHER’S CONFIRMATION OF ETHICAL STANDARDS COMPLIANCE
|
A. In this column, the researcher should confirm compliance with each ethical standard by entering Yes, No, or NA, and defending the response by providing supporting details. | B. Ethics reviewer will confirm compliance with each ethical standard in this column by entering “Confirmed” or provide a request for revisions. The researcher should enter responses/revisions using a font of a different color. |
| Sample: Will data be stored securely?
|
Sample response: Yes. Supporting details: Paper surveys will be stored in a locked file cabinet at the researcher’s home. Electronic files will be stored on the researcher’s password-protected computer and backed up on a password-protected hard drive. | |
| The first 13 questions apply to all studies (even when the researcher is not interacting with participants to collect new data).
Hover the mouse over the blue footnoted words to view extra tips and definitions.
|
||
| 1. Has each recruitment and data collection step[endnoteRef:1] been articulated such that risks/burdens can be identified? (Provide a bulleted list of recruitment and data collection steps in the brown column.) [1: ] | ||
| 2. Will the research procedures ensure privacy[endnoteRef:2] during data collection? Describe how. [2: Privacy risks might include unintended breach of confidential information (such as educational or medical records); being observed/overheard by others while providing data; or intrusion on the privacy of others who not involved in study (e.g. participant’s family).] | ||
| 3. Will data be stored securely[endnoteRef:3]? Describe how. [3: Secure data storage requires password protection on electronic files and locks for physical data.] | ||
| 4. Will the data be stored for at least 5 years? Describe how data disposal will occur. | ||
| 5. If participants’ names or contact info will be recorded in the research records, are they absolutely necessary[endnoteRef:4]? Describe why or clarify that data collection is 100% anonymous (which is preferable). [4: Note that consent forms do not require signatures if the participant can indicate consent by some action such as clicking on a link, returning a completed survey, etc.] | ||
| 6. Do the research procedures and analysis/writeup plans include measures to ensure that participant identities are not directly or indirectly[endnoteRef:5] disclosed? Describe how. [5: Participant identities might be “indirectly” and unintentionally disclosed if a researcher’s final research report fails to withold demographic details or site descriptions that might permit a reader to deduce the identity of a participant. So the researcher needs to think about which demographic descriptors are most important to collect and report, while ensuring that the identity of individual participants is protected. Also, the name of the site/organization is typically masked in scholarly research though in some cases, the organization can elect to publicize their name along with the research results.] | ||
| 7. Will confidentiality agreements[endnoteRef:6] be signed by anyone[endnoteRef:7] who may view data that that contains identifiers? (e.g., transcriber, translator) Submit a blank copy. [6: A sample confidentiality agreement can be found on the IRB website.] [7: Confidentiality agreements are required for transcribers or interpreters but not for the researcher or Walden faculty/staff who are automatically bound to confidentiality. Some professional transcribers/statisticians/etc address confidentiality in their work agreement and this is acceptable.] | ||
| 8. Is there a specific plan[endnoteRef:8] in place for sharing results with the participants and community stakeholders? Describe. [8: It is important that the format is audience-appropriate. Stakeholders may lack the time or inclination to digest a full research article or dissertation. Typically a 1 to 2 page summary or verbal presentation is most appropriate.] | ||
| 9. Have all potential psychological[endnoteRef:9], relationship[endnoteRef:10], legal[endnoteRef:11], economic/professional[endnoteRef:12], physical[endnoteRef:13], and other risks been fully acknowledged and described? In the brown column, provide a bulleted list of risks to participants, labeling which ones are minimal[endnoteRef:14] versus substantial[endnoteRef:15]. [9: Psychological risks include stress greater than what one would experience in daily life (e.g., materials or topics that could be considered sensitive, offensive, threatening, degrading).] [10: Relationship risks are present if the recruitment or data collection process are likely to alter the existing dynamics between the researcher and participant (who may be coworkers or have some professional relationship), among participants (if they know one another), or between the participant and the participant’s friends, coworkers, or family members.] [11: Legal risks are present if data collection might result in a participant’s disclosure of violation of laws.] [12: Economic/professional risks are present if data collection could result in the participant disclosing violation of workplace policies, disagreement with leadership decisions, poor work performance, or anything else that could be damaging to the participant’s position, professional reputation, promotability, or employability. ] [13: Physical risks are not common in social science research but would involve risk of serious physical injury.] [14: Minimal risk is defined as follows in U.S. federal regulations: “that the probability and magnitude of harm or discomfort anticipated in the research are not greater in and of themselves than those ordinarily encountered in daily life or during the performance of routine physical or psychological examinations or tests.” ] [15: Substantial risk is acceptable as long as adequate preventive protections are in place.] | ||
| 10. Have the above risks been minimized as much as possible? In other words, are measures in place to provide participants with reasonable protection from loss of privacy, psychological distress, relationship harm, legal risks, economic loss, damage to professional reputation, and physical harm? In the brown column, explain how each risk will be minimized. | ||
| 11. Has the researcher proactively managed any potential conflicts of interest[endnoteRef:16]? Describe how. [16: A conflict of interest is caused when the researcher has some sort of dual role in the research context, such as being a teacher, therapist, investor, business-owner, manager, etc. Conflict of interest must be managed to ensure that the research reveals “truth,” not just the outcome that the researcher might desire to see due to their other role. The simplest way to ensure this impartiality is to conduct research OUTSIDE of one’s own context but other methods are possible (e.g., using anonymous data collection to encourage honest responses).] | ||
| 12. Are the research risks and burdens[endnoteRef:17] reasonable, in consideration of the new knowledge[endnoteRef:18] that this research design can offer? Describe why. [17: All research activities place some degree of burden on the participants by asking the participants to share personal information, volunteer time, and assume risks.] [18: Examples of “new knowledge” include: effectively addressing a gap in the literature, generating new theory, enhancing understanding of a phenomenon, assessing effectiveness of a particular professional practice, addressing a local practical problem via data analysis.] | ||
| 13. Is the research site willing[endnoteRef:19] to provide a Letter of Cooperation granting permission[endnoteRef:20] for all relevant data[endnoteRef:21] access, access to participants, facility use, and/or use of personnel time for research purposes? (Note that some research sites will only release data if a more formal Data Use Agreement is in place, often in addition to a Letter of Cooperation.). State whether you will be obtaining written site approval before or after Walden IRB approval. [19: If a site requires the researcher to obtain Walden’s IRB approval before they can provide their written approval, that’s fine. (Walden can issue a “conditional IRB approval” letter to the researcher and then Walden’s IRB approval will then be finalized once the Walden IRB receives the community partner’s letter of cooperation.)] [20: No Letter of Cooperation is required (a) if the researcher will simply be asking organizations to distribute research invitations on the researcher’s behalf, or (b) if the researcher is using only public means to identify/contact participants. ] [21: Note that when medical, educational, or business records would be analyzed or used to identify potential research participants, the site needs to explicitly approve access to data for research purposes (even if the researcher normally has access to that data to perform their job).] |
The post ETHICS SELF-CHECK APPLICATION FOR IRB APPROVAL appeared first on Infinite Essays.
ETHICS SELF-CHECK APPLICATION FOR IRB APPROVAL
"If this is not the paper you were searching for, you can order your 100% plagiarism free, professional written paper now!"